Home / Mumbai / Mumbai News / Article /
Mumbai: FDA boss bats for change in system to recall drugs
Updated On: 28 November, 2022 07:55 AM IST | Mumbai | Suraj Pandey
Agency’s efforts call back a particular injection, following a patient’s death due to suspected adverse drug reaction, get poor response
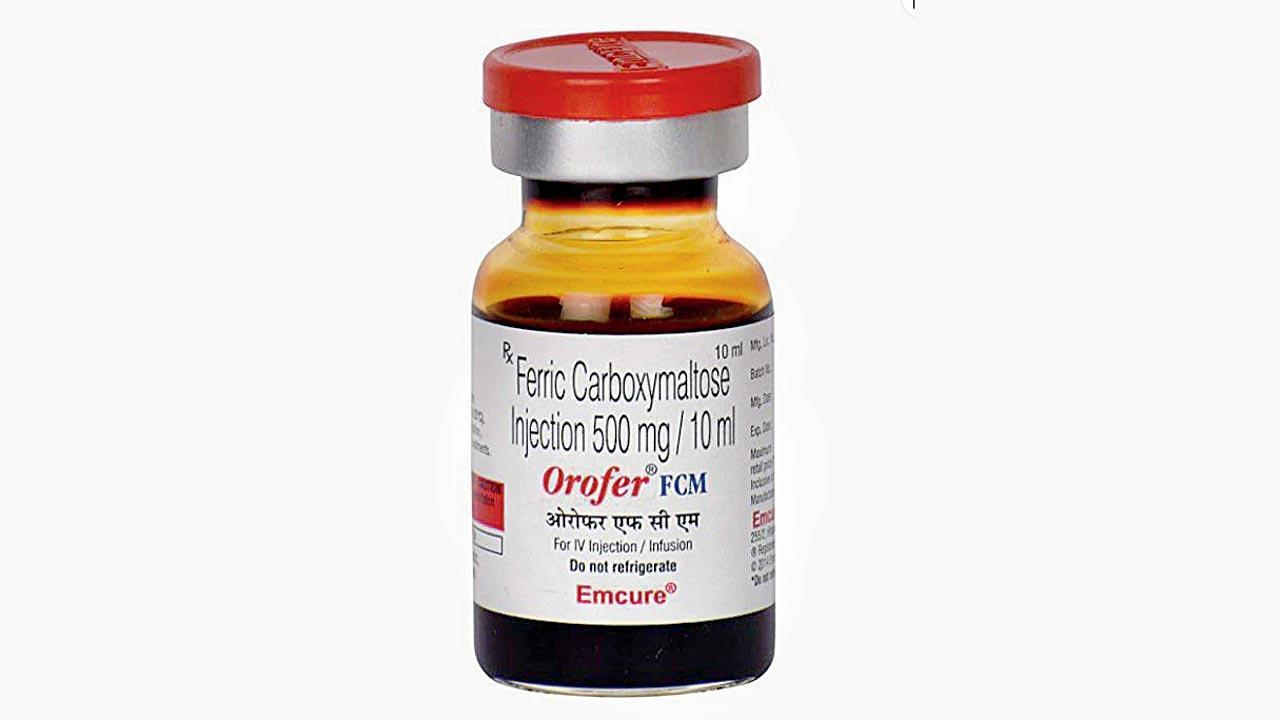
Emcure’s Orofer FCM (ferric carboxymaltose 500 mg/10 ml) is given to patients to increase haemoglobin
Maharashtra's Food and Drug Administration officials have expressed dismay after the agency’s efforts to pull back an injection from the market elicited a poor response, stressing that it was time to change the system of recalling drugs. Six days ago, the FDA ordered that Orofer FCM (ferric carboxymaltose 500 mg/10 ml), given to patients to increase haemoglobin, be recalled following a death due to a suspected adverse drug reaction.
So far, only 4 per cent of the 11,000 vials in the state have come back to the FDA.
How do you like the new new mid-day.com experience? Share your feedback and help us improve.

